The Bharal – formerly known as Blue Sheep – is a goat-like caprid. It is designed for living in precipitous and rocky terrain and may well be one of the most abundant mountain ungulates, maybe second only to the chamois. [2]
Names
Bhutia (Sikkimese-Tibetan language): Nao [8]
Chinese: 岩羊, Yanyang [12]
English: Greater Bharal, Greater Blue Sheep, Chinese Blue Sheep, Himalayan Blue Sheep, Tibetan Blue Sheep [15]
French: Bharal de l’Himalaya [15]
German: Bharal [2], Blauschaf [15]
Nepali: Nervati [8], Nahur [2]
Hindi: Bharal, Bharar, Bharut [8]
Ladakhi: Na, Sna (female), Napo (male) [9]
Spanish: Capra azul del Himalaya [15]
Bharal is the technically correct common name of Hindi origin, while „Blue Sheep“, as the animal is often inappropriately called (see Bunch et al. 2009), is a reference to the bluish sheen in the coat. Nayaur appears to stem from the Nepali word nahur – a native name for this species. Pseudes and ois are Greek words, meaning false and sheep respectively. [2]
Other (putative ) scientific names
Ovis nahura [9]
Taxonomy / subspecies / similar species
Pseudois nayaur Hodgson, 1833 [15]
The evolutionary relationships between Bharal, true sheep and true goats is clear today. As far back as the 1960ies the general assumption was that the genus originated from common ancestral goat stock in the early Pliocene (Thenius and Hofer, 1960). [2] Then Hassanin et al. (1998) determined the molecular systematics of the subfamily Caprinae on basis of cytochrome b sequences and made clear that Bharal are closer to Wild Goats (Capra) and the Himalayan Thar (Hemitragus jemlahicus) than to Wild Sheep (Ovis). [66] Another hint to this relationship: Pseudois nayaur hybridises with domestic goats but not with domestic sheep. [15]
Subspecies: Several authors [1, 2, 12] distinguish between Tibetan Bharal (P. n. nayaur) and Chinese Bharal (P. n. szechuanensis). They quote a slightly smaller size and lighter colour for the Chinese Bharal, further smaller horn length and circumference, but greater tip-to-tip-length. But boundaries between Tibetan and Chinese Bharal distribution ranges seem to be unclear. [1] A recent study (Tan, S. et al., 2017) corroborates the validity of these subspecies. [13]
The Dwarf Bharal has been recognized as a full species by various authors (Wang and Hoffman 1987, Wang et al. 2000, Liu et al. 2007, Wang and Xie 2009, Groves and Grubb 2011). On the other hand molecular evidence suggests to see it as a subspecies of Pseudois nayaur (Feng et al. 2001, Cao et al. 2003, Zhou et al. 2003, Zeng et al. 2008, Tan et al. 2012) [6]
Furthermore a small population in the Helan Shan (Helan Mountains, Ningxia Province, China) at the northeastern end of the distribution area is believed to belong to a new, yet undescribed subspecies of P. nayaur. [11] Tan, S. et al. (2017) confirm this finding based on molecular data from cytochrome b and the mitochondrial control region sequences [13]. We contribute a chapter of its own for this putative subspecies: Helan Shan Bharal.
The same study mentions another population on the Pamir Plateau, Western Kunlun Mountains, Xinjiang, China. Bharal from this region and further west and south (Tajikistan and Pakistan) could be yet another subspecies. Like the Helan-Shan-Bharal, animals from the Pamir Plateau have been significantly differentiated [13]. Again we have compiled a chapter of its own: Pamir Bahral.
Distribution
The Bharal is found in Bhutan, China (Gansu, South Inner Mongolia [15], Ningxia-Inner Mongolia border, Qinghai, Shaanxi [15], Sichuan, Tibet, south-eastern Xinjiang Uyghur Autonomous Region, and northern Yunnan), northern India, northern Myanmar (Rabinowitz and Tun Khaing 1998), Nepal, northern Pakistan [6] and probably Tajikistan [6, 11].
China: Its distribution runs from western Tibet, southwestern Xinjiang (Schaller et al. 1987) where there are small populations in the mountains bordering the western edge of Aru Co, extending eastwards with in scattered populations throughout the autonomous region. It also occurs in southern Xinjiang, along the Kunlun Mountains and in the Arjin Mountains. It is present in most mountain ranges of western and southern Qinghai into eastern Sichuan and northwestern Yunnan, as well as in the Qilian and associated ranges of Gansu. The eastern extent of its current distribution appears to be in the Helan Shan, which form the western border of Ningxia Hui Autonomous Region (with Inner Mongolia). [6]
Bhutan: It is found throughout northern Bhutan above 4.000 to 4.500 m asl. [6]
India: Bharal are fairly continuously distributed in the northern Himalayan and Trans-Himalayan regions of India, although the extent of its eastern distribution along the northern border of Arunachal Pradesh is still unknown. They are relatively common in many areas of eastern Ladakh (Jammu and Kashmir), and in parts of Spiti and the upper Parbati valley in northern Himachal Pradesh (Fox et al. 1986, S. Pandey pers. comm.). Bharal are known to occur in the Govind Pashu Vihar Wildlife Sanctuary and Nanda Devi National Park, and near Badrinath (Uttar Pradesh), on the slopes of the Khangchendzonga massif (Sikkim), and in eastern Arunachal Pradesh. Very recently, Bharal presence has been confirmed in the northwestern corner of Arunachal Pradesh near its border with Bhutan and China. [6]
Nepal: It is fairly continuously distributed to the north of the Greater Himalaya from the border with India and Tibet in the extreme northwest, eastwards through Dolpo and Mustang to Gorkha district in north-central Nepal. It then re-occurs in Nepal in at least two isolated areas: in Lamobogar, and on the southwestern slopes of Kanchenjunga near the border with Sikkim (India) (Schaller 1977, Wegge 1991). These two are probably connected with more extensive populations across the border in Tibet. [6]
Pakistan: Its main distribution range in Pakistan includes the upper Gujerab valley, the upper Shimshal valley, and the area eastward from Shimshal pass (District Gilgit), including part of Khunjerab National Park (Schaller 1976; Roberts 1977; Rasool 1986; Wegge 1988, 1989; G. Tallone in litt. 1993). Outside these areas there is a single, recent observation of one animal from Khunjerab pass (R. Hess pers. obs. 1985). Earlier, Roberts (1977) mentioned its occurrence (with proof) around Shigar and the Baltoro glacier (District Baltistan), however, we have no actual information from this area. Roberts’ (1977) source for its occurrence in the Passu valley was a quote from Lydekker (1907) who mentioned “Hunza valley, near Passu”, but possibly meaning the Shimshal valley. The presence of the species in the Passu valley is not confirmed (D. Mallon pers. obs. in litt.). [6]
Tajikistan: Grubb (2005) indicates that Bharal can be found in Tajikistan, but documentation for this is not provided. [6]
Evidence of Bharal in Tajikistan was also published by Sapozhnikow (1983): Remains of an adult male were found by a geologist on Sarykol Range (South-western Pamir). [11] – Author’s note: Sarikol Range is at the eastern edge of the Pamirs on the border of Tajikistan and China.
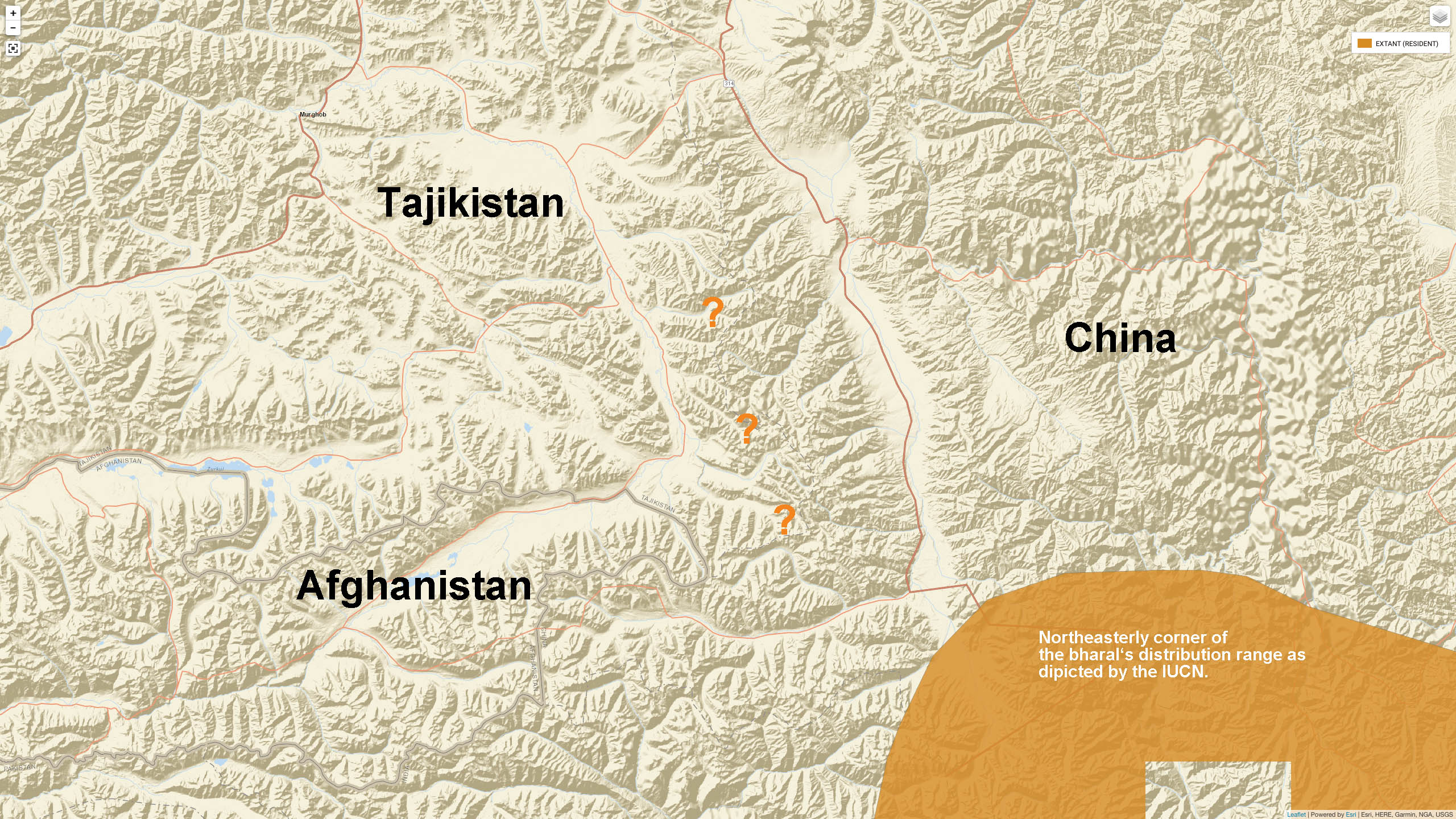
Presumed distribution area of Bharal in Tajikistan (marked with ???). Source: IUCN range map of Bharal – altered
Sapozhnikov also mentions some unknown ungulates repeatedly seen during aerial counts (from helicopter) in the Pamirs. It was speculated that these were Bharal. The data were neither confirmed later nor denied. – Sapozhnikow, G. N., 1983. The blue sheep (Pseudois nayaur) – new species of the USSR fauna. Proc. Sciences Tajik SSR, Bio. Dept., deposited in the VINITI (Information centre), 9 pp. (in Russian) [11]
Tan, S. et al. (2017) mention in their Bharal study the „Pamir Plateau“ as a sampling site. [13] The Pamirs lie mostly in Tajikistan, therefore this statement of place can be confusing. Tan’s actual sampling location lies in the Western Kunlun Mountains, Xinjiang, China.
Description
general appearance: The Bharal is stockily built, stout-legged and broad-chested, designed for living in precipitous and rocky terrain. The rather short legs lower the center of gravity and reduce heat loss. [2]
sheep and goat features: Bharal are sheep-like in the absence of a beard, any goat-like smell or calluses on the knees. Bharals do not possess, or have only rudimentary traces of, the ovis-typical glands, like inguinal glands (groin), preorbital glands (infront of eyes) and interdigital glands (between the digits). [2]. On the other side they resemble goats in having a broad, flat tail that is naked underneath, large dew claws and markings on the front limbs [5], as well as some skull features typical of goats [2].
length / head-body: 120-140 cm; males are over 30 per cent larger than females. [15]
shoulder height: 80-91,4 cm (males) [15]
weight: 60-75 kg (males), 35-55 kg (females) [15]
horn length: 38-76 cm (males), 10-20 cm (females) [15]
tail: 10 to 20 cm long [7], broad and flat with a bare central surface [12]
dewclaws: large [12]
karyotype: 2n = 54; one specimen from Gansu Province, China, had 56 [15]
longevity: 12-15 years in zoos [5], in the wild, animals older than 10 years are rarely encountered. A zoo animal died with 203/4 years. [7]
Colouration / pelage
overall body colour in winter: bluish-grey [15]; brown-grey with a bluish cast [4]
overall body colour in summer: brownish-grey [15]
front of neck and chest: black [15], lacking in females [9]
underparts: white [15], occasionally golden-brown [4]; a black stripe along the lower side of flank separates the body colour from the underside. The flank stripe is continuous with the black of the front of the hindlegs. [15] The flank stripe is more conspicuous in males and appears at age 4. [7] In females it is more grey instead of black. [15]
legs: The inside and back of limbs are white, fronts of all four legs are black [15]; sometimes black strips extend down the whole hindleg. [4]

The contrasty pattern of the legs is typical for Bharals as well as for true goats. Photo: Bürglin / Ladakh
head: dark-brown face-mask [4]
rump patch: white [7]

The backside of Bharals – with white on the backside of the hind legs coalescing with the white rump patch and black tail – is very conspicuous. Photo: Bürglin / Ladakh
tail: black [15]; two thirds black, underside naked [7]
Horns
Horns of males grow outward, then backward, and curve downward with the tips usually pointing slightly upward. [15] They are rounded [12] or more precisely roughly triangular in cross-section and show a ridge on the inner margin [2]. The surface is daintily wrinkled [12]. They resemble vaguely those of the Daghestan Tur. [2, 5] Horns initially show backward-pointing tips, then, from about 10 years of age in adult males, curving around in a semicircle. [4] Annual grooves are conspicuous. [7]

… same animal from behind: horn surface is smooth; latest annual grooves are clearly visible, but horn tips have worn off and oldest grooves have been lost. Photo: Bürglin / Mulhouse

Horn shapes of female Bharal: Another feature to distinguish between subspecies? Horns of female Chinese Bharal are said to be „nearly erect“, whereas in the Tibetan Bharal they are „much compressed and curving upwards and outwards in a scimitar-like fashion“. [2] Photos above: portraits of two females from Ladakh. Photos below: two Chinese Bharal with erect and lyre-shaped horns. All images by Ralf Bürglin

As in other caprinae species, horns do occasionally break off (animal in the middle) Photo: Bürglin / Taglangla, Ladakh
horns of young: Newborns do not show any horn edges. Horns start to develop after some weeks and reach 0,5 cm after two months. A male had 6 cm long horns after six months; and 10 cm long horns after 9 months. [7]

Female and young of the year in fall. Young approximately 5 month old, with poorly developed horns. Photo: Bürglin / Tsokar, Ladakh

Young of the year, approximately 7 months old; horns still not reaching the length of the ears. Photo: Bürglin / Mulhouse
horn length – in males: 38-76 cm [15], 82 cm [12]; – females: 10-20 cm [15]
tip-to-tip-spread: 57-82 cm (males) [15]; 59,6-73,4 cm [4]
horn basal girth: 23-34 cm [15]
basal diameter of horn core: greater 8 cm, usually greater 9 cm [4]
Habitat
Bharals occupy a variety of habitats across the region [6] with highly variable annual precipitation. [15] They inhabit open grassy slopes in high mountains from 2.500-5.500 m asl. Bharals are usually found near cliffs and similar escape cover, and generally avoid entering forests (Schaller 1977; the Helan Shan [see below] is a partial exception). This species is often able to maintain locally high densities in habitats otherwise inhospitable to other wild species. Often, these areas are close to domestic livestock (although generally at higher elevations, or in less productive habitats). They are very tolerant of environmental extremes from desert mountains in searing heat to windy and cold slopes (Schaller 1998). [6] Nevertheless Bharals choose better habitat if available.

If it was not for the rump patch, this young would also be hard to spot. Photo: Bürglin / Hemis NP, Ladakh
Habitats differ from area to area [15]:
In Xizang, China, they occur at 4.000-4.900 m in alpine sedge meadows and areas free of tall vegetation near talus or rough, precipitous terrain, and also in much lower, arid, hilly terrain.
In the Helan Mountains, China, an isolated desert mountain range in Inner Mongolia, Northern China, Bharals occur at 2.000-3.000 m in montane grasslands, woodland steppe, conifer forest, subalpine shrubland and meadows and had a preference for montane woodland steppe. In one protected study area of the Helan Shan availability of grass species in the genus Stipa probably most influenced foraging habitat selection.
A high-quality habitat in Nepal consisted of 29 per cent bare ground and 58 per cent grass cover. [15]
Food and feeding
Greater Bharals can obtain food resources from sparse, high-elevation vegetation. [15]

Consumed plant groups comprise grasses, mosses, herbaceous – including alpine – herbs and lichens. [6] Photo: Bürglin / Hemis NP, Ladakh, India

In some areas diets of Greater Bharals are dominated by graminoids. Photo: Bürglin / Taglangla, Ladakh
In Northern China, graminoids constituted 58,8 per cent of summer diets. Winter diets consisted of 36,7 per cent graminoids and nearly 50 per cent browse, including tree leaves. In Northern Nepal, Greater Bharal diet also was dominated by graminoids (51 per cent) and forbs and brows constituted about equal proportions. [15]

Ephedra was a much sought-after forage plant at the end of October in Hemis NP, Ladakh. Photo: Bürglin

Ephedra is a genus of gymnosperm shrubs, the only genus in its family (Ephedraceae). What looks like berries are actually female cones. Photo: Bürglin / Ladakh
Some plant taxa eaten by Bharal comprise: Arundinella sp., Danthonia sp., Festuca sp., Trisetum sp., Berberis, Cotoneaster, Ephedra, Lonicera, Juniperus, Poa alpina, Poa pratenis. [7]
Interspecific competition
Siberian Ibex: The ranges of Greater Bharal and Siberian Ibex (Capra sibirica) are sympatric in some areas. [15]
Tibetan Argali: In Ladakh, India, Greater Bharal and Tibetan Argali (Ovis hodgsoni) were spatially segregated due to habitat preferences and did not compete for forage. [15]
Livestock: Bharals also often share foraging areas with livestock, especially during summer. There can be substantial diet overlap with livestock, which can have a strong influence on the availability of forage. In Xizang, there was spatial overlap with livestock during summer and diet and habitat separation during winter. In another study the increased intake of browse by Greater Bharal in winter was due to the limited availability of graminoids because of their consumption by livestock in summer.
Sheep and goats: Bharal and domestic goats had the most similar diets, but differed in the proportions of the three forage classes consumed. In Diet and spatial overlap in one study was lowest between Bharals and domestic sheep and goats in winter.
Yak: Greater Bharals overlapped extensively with domestic Yaks because both used similar elevations and both consumed high proportions of browse. [15]
Mortality
Major mortality occurs in winter and spring due to climatic conditions and poor forage quality. Mortality rates of rams increases with age.

Sculls of dead ungulates – including Bharal – are collected by local Ladakhis and placed at waypoints and buildings. Photo: Bürglin / Rumbak, Ladakh
Table 1: Mortality rate of rams in different age groups [15]
| rams 1,5 to 4,5 years old | 0 to 7,4 per cent |
| rams 5,5 to 10,5 years old | 14,9 to 25 per cent |
| rams 11,5 to 15,5 years old | 40,7 to 100 per cent |
Percentage of rams that survived up to 8 years old was 64,7 per cent and that of rams that survived greater than 13 years was 7,8 per cent. On the Tibetan Plateau, over 80 per cent of males died between 4 and 11 years of age and at Dorpatan in Nepal, about 50 per cent of Bharal died between birth and 2 years of age. [15]
Predators: Bharals in the Manang District of Nepal (population about 1.000 animals) were the major prey of Snow Leopards (Panthera unica), which consumed 11-24 percent of the total number of Bharal annually. Other mammalian predators are Grey Wolves (Canis lupus), Leopards (Panthera pardus) and Dholes (Cuon alpinus) [15] and Himalaya Red Fox (Vulpes vulpes montana) [7]. But large predators have been extirpated in many areas [15]. Eagles (Aquila sp.) may lift lambs [9]. Matschei (2012) includes Steppe Eagle (Aquila nipalensis). [7]

Immature Steppe Eagle: The biggest specimens (80 cm) are about the size of Golden Eagles (88 cm). Both species are known to prey on Bharal lambs. Photo: Bürglin / India
Breeding
sexual maturity: 1,5 years, though males are not fully sexually active until age 7 [6]; most females give birth at age three. [15]
breeding season: December to January [6, 15]; in one study area in Nepal: mid-December to late January, with a peak in early January [15]
mixed groups: Males join females to form mixed groups and actively court females with a variety of courtship displays. Dominant males, older than five years old, do most of the mating. [15]
activity: Rams are envolved in combats for dominance over females. They either rear up on their hind legs and, leaning forward, crash horns (goatlike) or occasionally race towards each other head-on (sheeplike). [10]
estrus: lasts one to four days with a peak of about 20 to 36 hours. The estrous cycle lasts about 25 days and females recycle if not impregnated. [15]
gestation: 160 days [6, 15]
lambing season: May to June [15]; most of the Tibetan Plateau: mid-June [6]; one study area in Nepal: commenced on 23 May and probably peaked in mid-June, but births can extend into July [15]
young per birth: single lambs (rarely twins) [6, 15]
weaning: after six months [6]
creches: larger numbers of nursing young temporarily associated with a few females, have been documented. [15]
Activity pattern
active throughout the day [6]; Greater Bharals are diurnal and are most active before 9:30 h and after 16 h, but also feed about midday. In Nepal, during rut in December, Greater Bharal were least active during the warmest period of the day (13:30 to 15:00 h) and activity increased in late afternoon. In May to June, activity peaked later in the afternoon. [15]
Movements, home range and social organisation
Population parameters vary widely among areas. Greater Bharals are usually gregarious [15]. Sentinels watch out for predators. [6] Bharals have a squirrel-like „chirrt“ alarm call [8], a warning hiss or loud sharp alarm whistle. [9]
group size: Bharals live in small to rather large herds (300 individuals in one herd have been observed). [6]
On the Tibetan Plateau, female herds averaged 14,5 individuals and mixed herds 23,2 individuals. [15]
In the Manang District, Nepal, mean group size was 15,6, with smaller groups in winter than other seasons probably due to lower forage abundance and quality. [15]
Herds in the Helan Mountains consisted of an average of 4,2 individuals in winter and 5,6 in spring and summer. [15]
composition of mixed herds before and during rut: In one area in Nepal, 66 per cent of males were in mixed herds before rut and 80 per cent during rut. [15]
densities: In the Helan Mountains region in Northern China densities ranged from 3,6 ind/km² in spring to 4,6 ind/km² in summer of the following year. In better-quality habitats in other areas of China, densities were 3,6 to 5,3 ind/km². [15]
home ranges: were 3 to 7 km² for males and 2,5 to 5 km² for females in winter.
sex and age group ratios: per 100 females were 93,7 males in spring, 126,9 in autumn and 91,7 in winter. The population consisted of 34 per cent adult males, 32 per cent ewes, 15 per cent yearlings, and 19 per cent lambs. Mother-young groups were most numerous in spring and male-female groups were present only in winter. Adult males and females are not always segregated in separate herds during the non-mating period. [15]
lamb per ewe ratios: were 52,5 per cent in spring, 70 per cent in autumn and 55,4 per cent in winter. [15]
Yearling per 100 ewe ratios: were 32,9 in spring, 59,9 in autumn and 38,1 in winter. [15]
Yearling to female ratios: on the Tibetan Plateau usually were from 26:100 to 40:100. [15]
Status
The IUCN Red List classifies the Bharal as „least concern“ in view of its wide distribution, presumed large population, and because any decline is probably much less than the rate required to qualify for listing in a threatened category. [6]
Range-wide population estimates are unavailable. But taken together the estimates below, total population sizes could be roughly 47.000 (almost certainly a very conservative estimate) or as much as 414.000 (probably an overestimate). Bharals live almost entirely in steep, mountainous terrain. It is probably absent from broad areas devoid of steep topography (e.g., basins). However, this does not necessarily mean populations are fragmented, because it appears to be present in virtually all appropriate mountainous terrain from the Qilian Mountains in the north (Gansu and Qinghai provinces) to the Himalaya in the south. [6] The Greater Bharal is probably the most numerous ungulate in western China. [15]
China: Most extrapolations of population size in China are based on insufficient justification, and should be viewed with caution. However, in marked contrast to other species, field investigations of subjective and anecdotal reports of P. nayaur invariably confirm the presence and general abundance of this species. Thus, although specific figures in Chinese literature may be inaccurate, all evidence suggests that P. nayaur is widespread and relatively abundant within appropriate habitats within Chinese portions of the Tibetan plateau.
In Western China, P. nayaur was said to be “relatively abundant” (Shackleton 1997, Wang 1998), with various (questionable) local density estimates, and a total population greater than 10.000. The bulk of these 10.000 were an extrapolation for the Arjin Shan Nature Reserve, which is probably an overestimate. However, numerous areas within the current range were not included in this estimate, so the amount of 10.000 within the western portion of the Chinese range is not necessarily out of the question (Schaller 1998 guessed that at least 10.000 inhabited the Chang Tang Nature Reserve in western Tibet). Schaller (1998) cited an estimate by Pu (1993) of 34.500-49.600 in “western Tibet”, which includes but is larger than the Chang Tang Nature Reserve.
Further east within the Tibetan plateau, Wang (1998) cited a Chinese extrapolation of over 1.200.000 in Qinghai province (of which 267.000 were in “eastern” Qinghai), which is probably a considerable overestimate. Schaller (1998), in discussing the estimate by Pu (1993, see above), noted that it is likely that greater than 20 to 30 percent of the entire Chinese population lives east of “western Tibet”. If there are 30.000 to 50.000 in “western Tibet”, there may thus be roughly from 43.000 to 62.500 (using the derivation from Pu 1993), to 200.000 to 400.000 (using extrapolations from Wang 1998) within the entire Chinese portion of the Tibetan Plateau.
In the Helan Shan (Inner Mongolia), Ren et al. (1999) and Lü et al. (2000) estimated roughly 5.000 to 9.000 Bharal, but their extrapolations were based on non-randomly placed transects. [6] Other estimates quote even higher numbers for this area: 10.000 [15] Liu et al. (2005) have continued to monitor this population, which, regardless of methodological problems in estimating its size, is clearly dense and in no danger. [6]
India: Within India, Shackleton (1997) cites an estimate produced by Fox et al. (1991) of “a minimum of 11.000” within Ladakh.
Nepal: Shackleton (1997) reports a “conservative estimate of 10.000 animals”, although Schaller (1998) included a table suggesting 1.947 to 2.561 in areas counted within Nepal. Pakistan: Shackleton writes that, “according to written reports [Wegge 1988, Rasool 1990], the species is not as rare as previously thought … the most recent estimates indicated between 2.000 and 2.500 animals in 1992.”
Bhutan: No estimate available [6]
Threats
Competition with livestock has been suggested and probably occurs (Shrestha et al. 2005), but in general, habitat loss due to livestock threat is minor, and P. nayaur can sustain a reasonably large population under current and likely future scenarios with regard to the livestock industry. This is because reasonably large populations of the species can be supported entirely within high elevation, steep, or excessively rough/rocky terrain that is used sparingly if at all by domestic livestock. Livestock does exist near P. nayaur subpopulations, and in specific situations there may be conflicts. However, in general, it appears that even relatively intensive livestock grazing does not prohibit healthy populations of P. nayaur. This is in marked contrast to livestock’s effect on some other wild species. [6]
No guns, no poaching: Bharals are hunted for food. But because access to Bharal habitat is usually difficult and individual body-size is relatively small, market-based poaching for meat appears to be rare, at least in the northern portions of the Tibetan plateau. Locally-based, subsistence poaching has occurred frequently in recent years, and no doubt continues. However, China has recently begun implementing a new policy of confiscating all guns from nomadic pastoralists, and observations during 1998 to 2002 in Gansu and Qinghai provinces corroborate this (R. Harris pers. obs.). Thus, local herders now have little technology with which to poach Bharal for meat. Within Nepal, in some areas religious beliefs protect Bharal from poaching, [6]
Sarcoptic mange: An outbreak was reported among P. nayaur in extreme northern Pakistan, and has markedly reduced abundance locally. This may yet be a cause for concern range-wide, but similar reports from elsewhere within the species’ range have not surfaced (Woodford 2003, 2006). [6]
The expansion of grazing areas for domestic yaks could become a problem in Bhutan, although Bharals seem not to be in immediate danger there and numbers of Bharal increase. However, heavy grazing by both species is damaging the fragile, high alpine vegetation, so Bharal population trends may be reversed in the future. [6]
Localized over-hunting, including that by the army in remote outposts is a major threat in India. [6]
Limited range and low numbers make Bharal vulnerable to poaching and habitat loss in Pakistan. When access to the Shimshal valley is opened by construction of a jeep road, the species will most probably come under increased hunting pressure. [6] (According to geo.tv the road into the valley opened in 2004.)
Conservation
In China, Bharals are present within a number of large protected areas:
– Chang Tang Nature Reserve (Schaller and Gu 1994)
– Medoq, Qomolangma and Zayu Reserves (Tibet)
– the Arjin Mountain Reserve (Achuff and Petocz 1988, Butler et al. 1986, Gu 1990)
– and Taxkorgan Reserve (Schaller 1977; Xinjiang)
– Kekexili and Sanjiangyuan Nature Reserves in Qinghai
– Yanchiwan and Qilian Nature Reserves (western Gansu)
Some of these protected areas truly limit human disturbance, some have been demarcated where human disturbance was already quite low (generally because inhospitable climate makes production for human purposes marginal at best).
Other protected areas exist on paper only. Of note, however, is that, at least in China, Bharals appears able to persist at reasonable densities in the absence of protected areas. [6]
In Bhutan, Bharal are known to occur in Jigme Dorji National Park. Conservation measures proposed by Wollenhaupt (1989) included, using input from local inhabitants, establishing integrated alpine forest and grassland reserves in high elevation areas where demand for domestic yak grazing is considerable and deforestation and degradation of alpine areas occur (Wollenhaupt 1989). [6]
In India, Bharal occur in several national parks and many other protected areas in northern India (Fox 1987; Fox et al. 1986, 1991; Gaston et al. 1981, 1983; Green 1987; Pandey in prep.; Singh et al. 1990) including:
Jammu and Kashmir – Hemis National Park and Sabu Chukor Wildlife Reserve; Himachal Pradesh – Great Himalayan and Pin Valley National Parks and Chital, Daranghati, Kais, Kanawar, Lippa Asrang, Rakshum, Rupi Bhaba, Sangla Valley (includes previous Rakcha-Chitkul WS), Sechu Tuan Nala, and Tirthan (locally threatened) Wildlife Sanctuaries; Uttar Pradesh – Nanda Devi and Valley of Flowers National Parks, and Govind Pashu Vihar and Kedarnath Wildlife Sanctuaries; Sikkim – Khangchendzonga National Park; and their unlikely presence in Namdapha National Park of northeastern Arunachal Pradesh needs to be checked.
Conservation measures proposed for India:
1) Establish the proposed Changtang, Gya-Miru, Karakoram, and Lung Nag Wildlife Sanctuaries (Jammu and Kashmir), and the extensions to the Great Himalayan National Park and the proposed Srikhand National Park (Himachal Pradesh)
2) Determine if Bharal occur along the northern border of Arunachal Pradesh
3) Make the control of illegal hunting of this species the primary management priority to maintain viable populations under current land use regimes
4) Monitor changes in livestock grazing practices that could affect competition with Bharal [6]
In Nepal, Bharal are found in Shey-Phoksundo National Park and in the Annapurna Conservation Area. It is the main big game species in the Dhorpatan Hunting Reserve.
Conservation measures proposed for Nepal:
1) Maintain the current, closely controlled legal hunting program
2) Consider a regulated program of sustainable, low-level subsistence hunting by local villagers in some areas.
3) Steps should be taken to halt or reverse the habitat destruction caused by livestock grazing in the Bharal’s natural habitat. [6]
In Pakistan Khunjerab National Park contains a large portion of the country’s total population of Bharal. It occurs in no other protected area.
Conservation measures proposed for Pakistan:
1) Extend the boundaries of Khunjerab National Park eastwards.
2) Address problems faced by local people whose livelihoods would be affected by this extension. The management plan originally proposed for Khunjerab National Park did not do this, but this is necessary before its conservation recommendations can be implemented. The measures taken in the Bar valley project initiated by WWF-Pakistan and similar projects could serve as useful models.
3) Determine what detrimental influences the new road to Shimshal will have in the Park’s modified management plan. [6]
Trophy hunting
International (“trophy”) hunting for P. nayaur occurs (or occured), but only in four to five designated areas in China, Nepal and Pakistan. [6]
China: P. nayaur is categorized as a Class II protected species under China’s National Wildlife Law of 1988. Generally hunting of P. nayaur is legal only via permit obtained by provincial wildlife offices. In practice, such permits are provided only for foreign trophy hunters. Thus, hunting of P. nayaur is essentially illegal in China. International hunting in China – including for Bharal – was suspended in 2006, and as of summer 2014 had not yet been re-established. [6]
Pakistan: The Pakistani programs are conducted in community conservancies within the Moutain Areas Conservancy Project (MACP). Six permits for the 2010/2011 season were issued by he Wildlife Department of the Province of Gilgit-Baltistan. The expansion of the Pakistani trophy-hunting programs is hampered by unresolved issues between the Khunjerab National Park administration and the communities where hunting is conducted. The number of rams taken in these programs are low and do not have any impact on population structure and development. [2]
Nepal: Bharal hunting programs exist in the Dhorpatan Hunting Reserve; the authorities are also contemplating opening the Kanchendzonga Wildlife Reserve (12 to 14 permits for the 2012/2013 season) as a community hunting area.
Ecotourism
Many mammal watchers mention and value Great Bharals, especially from Ladakh, India – see trip reports. It is the most frequently seen ungulate in this region. Pfister (2004) states that the best place to see it in Ladakh is Hemis National Park. [9] Menon (2009) quotes that the species is best seen in India at Nanda Devi NP (Uttaranchal). [8]
Literature cited
[1] Castelló, José R., 2016: Bovids of the World – Antelopes, Gazelles, Cattle, Goats, Sheep, and Relatives. Princeton University Press
[2] Damm, Gerhard R. and Franco, Nicolás, 2014: The CIC Caprinae Atlas of the World – CIC International Council for Game and Wildlife Conservation, Budakeszi, Hungary in cooperation with Rowland Ward Publications RSA (Pty) Ltd., Johannesburg, South Africa
[3] Dickson, Barney; Hutton, Jonathan and Adams, Bill, 2009: Recreational hunting, conservation, and rural livelyhoods – science and practice. Blackwell Publishing, Ltd
[4] Groves, Colin and Grubb, Peter, 2011: Ungulate Taxonomy. The John Hopkins University Press
[5] Grzimek, Bernhard (Hrsg.), 1988: Grzimeks Enzyklopädie, Säugetiere, Band 5. Kindler Verlag, München
[6] Harris, R. B., 2014. Pseudois nayaur. The IUCN Red List of Threatened Species 2014: e.T61513537A64313015. Downloaded on 18 June 2018.
[66] Hassanin, Alexandre et al., 1998: Molecular Systematics of the Subfamily Caprinae (Artiodactyla, Bovidae) as Determined from Cytochrome b Sequences. Journal of Mammalian Evolution, Vol. 5, No. 3, 1998
[7] Matschei, Christian, 2012: Böcke, Takine & Moschusochsen. Filander Verlag
[8] Menon, Vivek, 2009: Field guide to Indian mammals. A&C Black Publisher Ltd., London
[9] Pfister, Otto, 2004: Birds and Mammals of Ladakh. Oxford University Press
[10] Schaller, George B., 1977: Mountain Monarchs – wild sheep and goats of the Himalaya. The University of Chicago Press
[11] Shackleton, D. M (ed.) and the IUCN/SSC Caprinae Specialist Group, 1997: Wild Sheep and Goats and their Relatives. Status Survey and Conservation Action Plan for Caprinae. IUCN, Gland, Switzerland and Cambridge, UK. 390 + vii pp
[12] Smith, Andrew T. and Xie, Yan, 2008: A guide to the mammals of China. Princeton University Press
[13] Tan, S.; Wang, Z.; Jiang, L.; Peng, R.; Zhang, T.; Peng, Q.; Zou, F., 2017: Molecular phylogeny and phylogeography of genus Pseudois (Bovidae, Cetartiodactyla): New insights into the contrasting phylogeographic structure. Ecol Evol. 2017 Jul 31; 7(17):7047-7057.
[14] Valdez, Raul, 1985: Lords of the pinnacles – wild goats of the world. Wild Sheep and Goat International, Mesila, New Mexico 88046
[15] Wilson, D. E. and Mittermeier, R. A. [eds], 2011: Handbook of the mammals of the world. Vol. 2. Hoofed mammals. Lynx Edicions, Barcelona.























