The Dwarf Bharal – also known as Dwarf Blue Sheep – is a goat-like caprid. It is much smaller than the Greater Bharal, has differently formed horns and a less conspicuous pelage pattern. One subpopulation has developed a unique feeding behaviour.
Names
Chinese: 矮岩羊, i yán yang [2], Ai Yanyang [13]
English: Dwarf Blue Sheep [16], Sichuan Blue Sheep [13, 16], Pigmy Blue Sheep [1]
French: Bharal nain [16], Petit Bharal [2]
German: Zwergblauschaf [7, 16], Zwergbharal [2]
Russian: карликовый голубой баран [2]
Spanish: Capra azul enana [1, 16], Bharal enano [1, 2]
Tibetan: Rong-na [2]
Other (putative) scientific names
Pseudois schaeferi [1, 2]
Taxonomy
Pseudois schaeferi Haltenorth, 1963, China, upper Yangtze Gorge, Drupalong, south of Batang [16]
The taxonomic status of the Dwarf Bharal has been debated intensively. Various authors (Wang and Hoffman 1987, Wang et al. 2000, Liu et al. 2007, Wang and Xie 2009, Groves and Grubb 2011) recognize it as a full species. Several others suggest on basis of molecular evidence to see it as a subspecies of Pseudois nayaur (Feng et al. 2001, Cao et al. 2003, Zhou et al. 2003, Zeng et al. 2008, Tan et al. 2012) [6].
Tan et al. (2012), who conducted a mitochondrial DNA study, cluster Dwarf Blue Sheep closely with some Blue Sheep from Sichuan Province. For them Dwarf Blue Sheep appear to be a subset of Pseudois nayaur szechuanensis. [15]
However because of striking morphological and biogeographical differences – much smaller size than Greater Bharal, differently formed horns, less conspicuous pelage pattern, separated distribution range – we here follow the IUCN Red List and consider the taxon Pseudois nayaur schaeferi.
Greater Bharal and Dwarf Bharal are separated by a forest zone that extends 1000 metres between the lower-elevation habitat inhabited by Dwarf Bharal and the higher-elevation habitat of the Greater Bharal. [16]
Groves (1978: 182) argued that genetic isolation and inbreeding, and environmental effects and malnutrition, are unlikely to account for an approximately 50 per cent size difference between the two forms, as was proposed by Allen (1939). [2]
Genetic analyses (concerning sequences of mitochondrial control regions) in four specimens of each showed an absolute difference of more than 12 per cent. If a rate of divergence of 6 to 12 per cent per million years is followed, then they separated 1 to 2 Ma (million years) ago (Feng et al., 2001). [4]
In further genetic analyses (concerning y-linked zinc-finger genes / ZFY intron sequences) the same authors found that, in contrast, ZFY were not completely separate, which suggests that the occasional Greater Bharal male may make his way through the forest belt and then, dominating Dwarf Bharal rams, mate with the dwarf ewes. [4]
Similar subspecies
The Greater Bharal is larger, with more distinct black markings on front of neck and chest and a wider black lateral band, and larger horns with a downward curve. Females of the two species are very similar. [1] The Dwarf Bharal is supposed to have more of a darkish colour than the Chinese Bharal. [2]

Comparison of Greater Bharal (left) and Dwarf Bharal – both animals are mature males: The difference in shoulder hight is about 10 centimetres (note the person who holds the dead Dwarf Bharal for scale), but male Dwarf Bharal weigh only half as much as their bigger cousins. It has moreover much smaller horns. Black markings are reduced and appear in this specimen only on the front of the limbs. Photo: Bürglin / Schäfer
Discovery
In 1934 the Dolan-Schäfer expedition (2nd Brook-Dolan Expedition, Academy of Natural Sciences, Philadelphia) discovered the Dwarf Bharal south of Batang, in the deep V-shaped gorge of the Jinsha River aka Jingsha Jiang River (upper reaches of Yangtze River) along a belt of dry, steep and rugged, rocky slopes between 2.600 and 3.500 metres. Ernst Schäfer collected 12 specimens of this unique animal (Schäfer 1937). Damm and Franko (2014) write that the Dolan-Schäfer expedition failed to name the Dwarf Bharal, which does not meet the facts.

German Zoologist Ernst Schäfer with chinese guides and two bagged Dwarf Bharal. Photo: Ernst Schäfer
In his book „Unbekanntes Tibet“ (engl.: Unknown Tibet) Schäfer has captioned his sixth chapter „Das Zwergblauschaf“ (engl.: the Dwarf Blue Sheep). On page 213 he writes: „Das Auffinden dieses Tieres wurde zu einem der größten Erfolge der Dolan-Expedition.“ (engl.: The discovery of this animal became one of the greatest successes of the Dolan expedition.) [11]
Actually two locals informed Schäfer about the species. He writes on page 215: „Zwei meiner Tibeter aus Batang behaupteten nämlich, dass in diesen gigantischen Felsschluchten Wildschafe vorkämen. Diese ähnelten den Blausschafen der Hochgebirge, doch seien sie viel kleiner …“ (engl.: Two of my Tibetans from Batang claimed that wild sheep would occur in these gigantic gorges. These resembled the Blue Sheep from the alpine areas, but they would be much smaller.) [11]
For whatever reason Schäfer’s report missed to find it’s way into the anglophone scientific literature. Only in 1963 did another german zoologist, Theodor Haltenorth, classify it as a subspecies of P. nayaur. Eventually Groves (1978: 182) raised the rank to the full species, P. schaeferi, based on „the distinctiveness and isolation of Dwarf Bharal, together with its geographic approach to the greater form“. [2]

Not a Dwarf Bharal: To this day – 2018-12-05 – there is no image of a life Dwarf Bharal available in the internet. All Wikipedia entries (9) use this photo of a dead animal from the Dolan-Schäfer-expedition, which actually shows a Greater Bharal – as you can see by the pronounced black markings on chest, limbs and flanks. Check also the labelling of the photo. Photo: Ernst Schäfer
Distribution
The Dwarf Bharal is found in China (Upper Yangtze Gorge in west Sichuan and adjacent parts of Tibet and north Yunnan) (Grubb 2005). Its primary range is in a narrow area along the Jinsha Valley (Golden Sand River [12]), which forms part of the upper reaches of the Changjiang (Yangtze) river. [6] The taxon is endemic to China. [13]
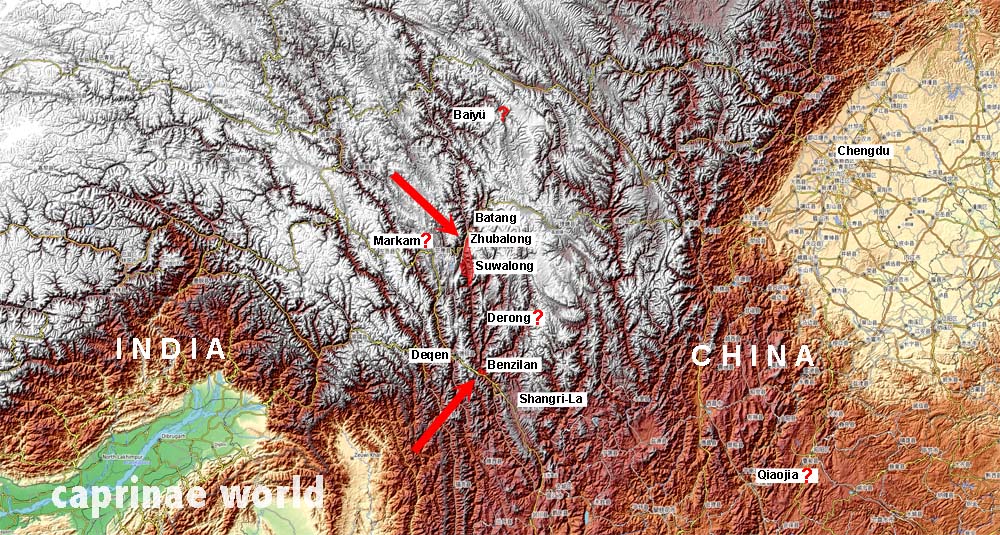
Actual and potential distribution areas of Dwarf Bharal along the upper Yangtze River Valley. Subpopulations are fragmented. Map basis: OpenTopoMap

View from the Jinsha River bent lookout near Benzilan. The slope between the viewpoint and the river – especially to the left hand side – is Dwarf Bharal habitat. Photo: Bürglin
Specimens have been reported from:
– near Batang, Sichuan [6] – between Zhubalong and Suwalong
– Baiyu, north of Batang – doubted by Wang et al. (2000) (believing these animals to be Greater Blue Sheep) [6]
– Derong, southwestern Sichuan – claimed to be found by local hunters [6]
– Markam, eastern Xizang – doubted by Wang et al. (2000) (believing these animals to be Greater Blue Sheep) [6]
– Luoni Mountains (aka Luoyi Shan or Luoyebai), northeastern Yunnan (Qiaojia County in Zhaotong Prefecture) – location is unconfirmed and could possibly refer to P. n. szechuanensis. [2]
– Rini Moutain, Baima Snow Mountain National Nature Reserve, Deqin county, northwestern Yunnan [3] – (It was believed that the status of Dwarf Blue Sheep in Deqin County is in some dispute [6]. Li De-Pin et al. (2013) confirmed the species for Rini Mountain. [3])
Description
general appearance: The Dwarf Bharal is smaller in body and horn size than its larger cousins, with rams not exceeding 40 kilograms [2] Males are only half the weight of the Greater Bharal! Females are less reduced it size. [4]
body length:
138 cm males – compared to 142 cm in male Chinese Bharal [2]
127 cm females – compared to 142 cm in female Chinese Bharal [2]
shoulder height:
80 cm [2]; 70-80 cm [16] in males – compared to 91,4 cm in male Greater Bharal [2]
weight: 28-35 kg [16]; 39 kg in males [2] – compared to 60-75 kg in the male Greater Bharal [16], 60 kg (male Chinese Bharal) [2]
– ca. 25 kg (females) [2, 15]; 25-35 kg (females) [2] – compared to 35-55 kg in female Greater Bharal [16], 39 kg [2]
horn length: 28-55 cm (males) – compared to 38-76 cm (males) in the Greater Bharal [16]
tail: 7 to 12 cm long [13]
karyotype: 2n = 54 [16]
Colouration / pelage
in general: drabber and more of a darkish colour than the Chinese Bharal [2]. The hair is shorter than nayaur and has less underwool. [4] New hairs bear dark terminal bands but these are short and typically wear off quickly, at least in the winter pelage. [2]
overall body colour in winter: drab (germ.: graubraun), with a silvery sheen; dark markings poorly developed [4, 15]

Pelage colour – hard to define: The general colour of this ewe and her young could be described as brownish (Photo taken at Rini Mountain in bright daylight at the end of october) … Photo: Bürglin

… probably the same animal (same location, same date) appears blueish while standing in the shade. Photo: Bürglin
overall body colour in summer: brownish-grey, with slightly yellowish tinge to the upper parts (Wu et al. 1990) [2]
black markings on head, neck and lateral flank stripe: only weakly expressed, except sometimes in old rams. [2]
underparts: white [4]; less white than in the Chinese Bharal [2]
legs: inside and back are white; darker than the body and maybe uniformly gray-black [2], dark overall [4]
rump patch: white [2]
Horns
Horns of males: Initially the horns run up and out, then the tips turn towards the body axis, but pointing upward and outward.
Compared to those of the Greater Bharal the horns are shorter and thinner [2]. They also do not form (downward sloping) semicircles. [4] They lack the (longer) inward curve characteristic of the horns of Greater Blue Sheep. [16]
Horns of females: They have small, straight horns [1]; they are very similar to those of female Tibetan Bharal. (At second glance) they slightly curve, first upward then outward. The cross-section is sub-oval (Wang and Hoffmann 1987). [2]
horn length: 28-55 cm (males) [16]; they rarely exceed 38-43 cm [2]
tip-to-tip-spread: 54,3 mm (average); 56,6 cm (maximum) – compared to Greater Bharal: 57-82 cm (males) [16]; 59,6-73,4 cm [4]
horn basal girth: 14-23 cm (males) [16]; 19-24 cm [2]
basal diameter of horn core: less than 8 cm – Greater Bharal: greater 8 cm, usually greater 9 cm [4]
Habitat
Dwarf Blue Sheep inhabit rugged valley terrain along the Yangtze River valley. They live among very steep rocky slopes between 2.700-3.200 metres asl. [6, 13], respectively 2.700-2.900 metres asl [16] and 2.050-3.300 metres asl at Rini Mountain [3].
General landscape pattern and separation of the two Bharal species:
The north-south running Jingsha Jiang River valley acts as a funnel for warm air from the south. The zone from the valley floor to about 3.200 metres receives little precipitation. Above 3.200 metres the v-shaped gorge expand into wide, glacier-carved slopes. This part of the landscapes receives higher precipitation, which in turn supports the growth of forests. The tree line is situated at about 4.500 metres, hence the forest zone spans about 1.000 metres and separates the Dwarf Bharal habitat altitudinally from the Sichuan Bharal (Pseudois nayaur sichuanensis) distribution range in alpine habitat at higher altitudes above 4.500 metres. [2]

Surrounding mountains as seen from a lookout at Rini Mountain. Depicted is the forest belt that separates Dwarf Bharal habitat from Greater Bharal habitat. Photo: Bürglin
Generally, both species avoid the forest. It is said that Dwarf Bharal only occasionally range into the lower fringes and clearings of the forest zone and Sichuan Bharal only enter the upper outer edges of the forest in winter. Therefore, ecological isolation and a likely lack of gene flow between these almost parapatric populations (ranges do not significantly overlap but are immediately adjacent to each other) was suggested by Feng et al. (2001).
In the northern part of the distribution area, near Batang, Dwarf Bharal occupy the rocky inclines below the forest belt and above the landslide slopes immediately rising from the river bed. The landslide slopes are used for agriculture by the local residents (Schäfer 1937). Towards the north, the steep erosion valleys diminish and the forest zone expands to the river bottom, thus limiting the distribution range of Dwarf Bharal towards the north. (Schäfer 1937) [2]
Common plant species include grasses (Cymbopogon distans and Themeda hooderi), low shrubs (Berberis spp., Rosa spp., Cotoneaster spp., Cladrastis spp., Ephedra spp., and Rhododendron spp.), and clubmoss (Selaginella sanguinolenta) (Wang et al. 2000). [6]
Rini Mountain, in the southern part of the distribution area, is separated from the main range by a low saddle and a mixed broadleaved-conifer forest belt (above 3200 m) of mainly Yunnan pine (Pinus yunnanensis), Chinese cypress (Cupressus duclouxiana), and the oak Quercus guajavifolia. The lower and midslopes are rocky steep and unstable; massive cliffs, are interspersed by small pockets of sparse grassland, shrubland, scurb, and loose scree deposits. The scrub and shrubland vegetation within the main habitat (2050-3300 m asl) mainly comprises the shrubs Chinese Chaste Tree (Vitex negundo var. microphylla), the Sandalwood (Osyris quadripartita), Sophora davidii, and the introduced Prickly Pear Cactus (Opuntia ficus-indica). Along the river grow scattered Pistacia (Pistacia weinmanniifolia). [3]

Main Dwarf Bharal habitat at Rini Mountain: left of the river; beyond the gully that comes down from the left hand side. Photo: Bürglin
The area is sparsely populated by sedentary agropastoralists who raise goats (Capra hircus), yak (Bos grunniens), and grow barley. Livestock are grazed within the habitat of the Dwarf Bharal. [3]
The climate at the side is arid and warm, with mean annual temperatures from 14° C to 17° C, and with monthly means ranging between 24° C in July to 8° C in January (Jin 2002). Total annual precipitation is less than 350 millimeter, with 90 per cent occurring as rainfall during the wet season (May to September). Annual mean evaporation is 3000 to 4000 millimeter (Jin 2002 ). Snow occurs regularly in winter down to ca. 3200 metres asl (near lower treeline). [3]
Habitat use: Dwarf Bharal use all five habitat types: cactus scrub, scree cactus scrub, scree scrub, cliff shrubland, and shrubland. Although more than 60 per cent of the observed times is spent in cliff cactus scrub.
The habitat use between age-gender groups among the five major habitat types at the site is significantly different: Adult males use the cliff cactus scrub habitat more, while the juveniles and adult females use the scree scrub habitat more often. [3]
Food and feeding
Dwarf Bharal are known to consume more than 20 species of plants (Wu et al. 1990); according to the observations made by Wang et al. (2000), they feed primarily on grasses – e.g., Flacid Grass (Pennisetum flaccidum) and Yellow Foxtail (Setaria glauca), though other plants are also eaten. [6]
17 food species of Dwarf Bharal were identified at Rini Mountain, Benzilan, Yunnan; though only 6 species made up nearly 90 per cent of the diet: Opuntia ficus-indica, Themeda triandra, Festuca durata, Polygonum thunbergii, Elsholtzia cypriani, and Excoecaria acerifolia. [3]
Number 1 food species: The exotic succulent prickly pear cactus (O. ficus-indica) alone accounted for almost half of the observed feeding occurrences. By foraging class, the diet was dominated by succulents (43 %), with graminoids (18 %), herbs (17 %), and woody plants (21 %) making up about equal proportions. Lycopods (Selaginella sp.) accounted for less than 1 per cent of the diet. [3]
Cactus use: Dwarf Bharal may have shifted their diet to include cactus as a result of low graminoid (grass) abundance or quality or in response to a new resource. Numerous animal species are reported to browse Prickly Pear Cactus (Opuntia spp.), though not always without ill consequences. [3]
Mouth injuries and chronic infections are common (Chavez-Ramirez et al. 1997). Some livestock will only browse the cactus when starving or preferentially browse where spines have been removed (Chavez-Ramirez et al. 1997). [3]
Horns used to remove cactus spines: Li De-Pin et al. (2013) also recorded adult sheep frequently raking their broad (ca. 9 cm diameter; Liu et al. 2007b ) horns along the broad cladodes (flattened photosynthetic stems of Cactaceae) effectively removing spines from the fleshy cladodes before feeding. The juveniles with less-developed horns were never observed exhibiting this behavior and only rarely ate the cactus, usually targeting new growth. While the behavior of rubbing cacti to remove the spines is known in Caprinae species, e.g., Bighorn Sheep (Ovis canadensis) in North America (Warrick and Krausman 1989, Tarango et al. 2002 ), no records exist for this behavior in Bharal (Pseudois sp.), and no other native plant with a similar morphology or defense mechanism exists within the range of Pseudois schaeferi or P. nayaur. In the Li De-Pin et al.-study (2013), domestic yaks and goats were observed to eat O. ficus-indica, as was the Chinese goral (Naemorhedus griseus), on one occasion, though none displayed the behavior of using their horns to remove the spines. [3]
Allover meaning of Prickly Pear Cactus for Dwarf Bharal: The winter diet of the Dwarf Bharal at Rini Mount has undoubtedly changed with the invasion of the highly palatable exotic succulent Opuntia ficus indica. This cactus is a long-domesticated crop species estimated to have originated in Central Mexico (Griffith 2004). It was first introduced in China around 1645 as a hedge and for its edible young joints and fruit (eFloras 2008). No other records exist of P. schaeferi or its close relative Pseudois nayaur consuming O. ficus-indica, although this cactus extends as far north as Batang. Li De-Pin et al. (2013) believe that the diet of no other wild population of threatened or endangered ungulate depends so much on an invasive exotic species. [3]

Kid and cactus: Note the ragged look of the cactus, which results from a heavy feeding pressure, although young have not been documented feeding on cactus. Photo: Bürglin
Predators
Principal Predators are Grey Wolves (Canis lupus), Leopards (Panthera pardus) and Dholes (Cuon alpinus) [2, 15] and large raptors [2]

Raptors are said to prey on Dwarf Bharal. This one, photographed at Rini Mountain, is possibly a Golden Eagle (long tail, base of wings narrower, wings held in shallow v, mountainous environment) Photo: Bürglin
Breeding
sexual maturity: 1,5 years; males may take seven years to reach full size (Wang and Hu 2004), and it is unlikely that rams participate in the mating process before that age. [2]
breeding season: November and December [16]
mixed groups: Males sometimes form all male groups or mix with females and young. [6]
gestation: 160 days [2]
lambing season: May to June [2, 15];
young per birth: single lambs, rarely twins [2]
weaning: after six months [2]
Activity pattern
Dwarf Bharal feed and rest alternately throughout the day. [13] Feeding peaks in summer occur at 6 to 9 am, 10 to 11 am and 4 to 8 pm, and a bedding peak at 11 am to 15 pm. Adult males spend more time moving and in other activities; females spend more time bedding. The species is characterized as timid and wary. [16]
Group sizes are 2 to 15 with an average herd size of 6,2. In another study group sizes were 2-10 animals [16]. Local hunters reported that the previously observed group size ranges of 10 to 36 have dropped to 3 to 8 animals in recent years. [2] Contrary to Schäfer’s original reports (1937), neither Wu et al. (1990) nor Wang et al. (2006) ever observed solitary individuals. [6]
Population densities range between 0.5-1.0 individuals per square kilometre. (Wu et al. 1990, Wang and Wang 2003). [6]
Sex and age group ratios: In one study of 142 Dwarf Blue Sheep observed,
21 (14,8 %) were males,
31 (21,8 %) were females, and
11 (7,7 %) were young;
79 (55,6 %) were of undetermined gender.
The sex ratio was 67,7 males : 100 females;
the ratio of young to combined adult and yearling females was 35,5-100 : 100 in winter. [16]
In another study age group ratios were (of those whose sex and age were determined):
17 (31 %) were young,
39 (29,8 %) were subadults,
and 75 (57,2 %) were adults
The adult sex ratio was 70,4 males : 100 females. [16]
Status
IUCN classifies the species as „endangered“. [6]
Populations seem to have declined sharply in the 1950s and 1960s, probably because of poaching, habitat loss and competition with livestock (Wang et al. 2000). [2] The population in Batang County has undergone a decline estimated to be greater than 50 percent in 21 years [16]. Some authors believe that if effective protection measures are not adopted quickly, the taxon will disappear in the near future. [6, 12].
Hu (1998) believed there were approximately 7.000 individuals in the mid-1990s, although this seems difficult to square with the reports of only a few hundred dred by Wang et al. (2000) – see above. [2]
However, Liu et al. (2010), who studied group characteristics of Dwarf Bharal in Zhubalong Nature Reserve, Sichuan Province, (winter of 2007 and 2008, and spring 2009) observed a total of 73 groups including 451 individuals in the spring period, and 170 groups including 1.036 individuals in winter. Among the individuals observed, there were 607 adults (236 males, 271 females), 143 subadults and 286 kids in winter; and 274 adults including 115 males and 159 females, 49 subadults and 128 kids in spring.
The results of Liu et al. (2010) seem to coincide with Hu (1998) and may be interpreted that the population numbers of Dwarf Bharal are considerably higher than the „few hundred“ which are cited frequently in literature (Wu et al. 1990; Wang et al. 2000: Wang and Wang 2003; Smith and Xie 2008). [2]
Threats
The area is subjected to extensive livestock grazing and farming. Its small distribution renders it especially vulnerable to continued habitat degradation by unmanaged domestic livestock grazing and disturbance by pastoralists. [16]

At Rini Mountain livestock enter the Dwarf Bharal habitat and use the same resources, here a mineral lick (the roundish structure in the upper right corner is presumably a plundered wasp nest). Photo: Bürglin
Poaching also occurs and game laws are not enforced even within a protected area. Despite the small population and declining status, it is not formally on the national list of protected species in China. [16]

Dwarf Bharal at Rini Mountain are very wary. At mineral licks poachers can watch for an opportunity to get close. Photo: Bürglin
Conservation action
The subspecies is protected under Chinese national law as a Category II protected species under the wildlife protection law of 1988. Since the taxon was recognized as unique and important, a concerted effort has been made by scientists temporarily working in the area to educate local hunters. [6]
In 1995, a prefectural reserve covering 142.4 square kilometres (which was enlarged to about 300 km² in 2007) around Zhubalong near Batang was established for the protection of this species (Wang et al. 2000). However, many human activities such as mushroom gathering, livestock grazing, and illegal hunting continue to occur in the core zone and thus threaten the populations here (Wang et al. 2000). [6]
Conservation measures proposed:
1) Continue to examine its taxonomic status.
2) If future research alters our current understanding and shows it to be more appropriately considered a full species, it should be raised to a Class I species in the national protection list.
3) Surveys are essential to determine status and total distribution throughout its suspected range.
4) More protected areas need to be established. [6]
The creation of a community based conservation hunting program that economically benefits the rural population and caters to foreign hunters is another possibility, but only if a detailed management plan is developed that provides for trained personnel to monitor and enforce hunting regulations and a multispecies grazing program that provides for the sustainable use of rangelands. [16]
Trophy hunting
currently not a factor.
Very few western hunters have ever hunted Dwarf Bharal and even fewer heads have been recorded in the listings of Safari Club International (SCI) and Rowland Ward. Three heads reportedly taken in Batang county, with horns much longer than those given as representative by Wang and Hoffmann (1987) and also longer than the old rams collected by Schäfer during the Dolan-Schäfer expedition, give reason to suspect that these trophies actually represent P. n. szechuanensis. However there is no information at which altitudes these animals were collected. [2]
Opposition from locals: Just prior to the closure, the last foreign hunters in the area (Baiyu, Sichuan Province) met opposition from local groups, probably since Dwarf Blue Sheep receive protection because of local religious beliefs. Smith and Xie (2008) also report that Dwarf Bharal are protected by the Baiyu minority on religious grounds. [2]
Ecotourism
Not a factor at the moment, although at Rini Mountain near the town of Benzilan tourist facilities are already in place: At the upper edge of the Dwarf Bharal habitat is a lookout. Thousands of tourists stop here annually to see the bend of the Jinsha River – without knowing that the slope below is home of a chinese endemite, a species with one of the most restricted ranges.

Entrance to the Jinsha River bent lookout. Thousands of visitors come here, most of them not knowing that they are visiting Dwarf Bharal country. Photo: Bürglin
Literature cited
[1] Castelló, José R., 2016: Bovids of the World – Antelopes, Gazelles, Cattle, Goats, Sheep, and Relatives. Princeton University Press
[2] Damm, Gerhard R. and Franco, Nicolás, 2014: The CIC Caprinae Atlas of the World – CIC International Council for Game and Wildlife Conservation, Budakeszi, Hungary in cooperation with Rowland Ward Publications RSA (Pty) Ltd., Johannesburg, South Africa
[3] Li De-Pin et al.: Feeding differences in Dwarf Blue Sheep, Pseudois schaeferi (Cetartiodactyla, Bovidae) magnified by the expansion on an invasive plant species. Mammalia 2013; 77(2): 131-140
[4] Groves, Colin and Grubb, Peter, 2011: Ungulate Taxonomy. The John Hopkins University Press
[5] Grzimek, Bernhard (Hrsg.), 1988: Grzimeks Enzyklopädie, Säugetiere, Band 5. Kindler Verlag, München
[6] Huffman, B. & Harris, R. 2014. Pseudois nayaur ssp. schaeferi. The IUCN Red List of Threatened Species 2014: e.T18535A64313668. http://dx.doi.org/10.2305/IUCN.UK.2014-3.RLTS.T18535A64313668.en. Downloaded on 03 December 2018.
[7] Matschei, Christian, 2012: Böcke, Takine & Moschusochsen. Filander Verlag
[8] Menon, Vivek, 2009: Field guide to Indian mammals. A&C Black Publisher Ltd., London
[9] Pfister, Otto, 2004: Birds and Mammals of Ladakh. Oxford University Press
[10] Schaller, George B., 1977: Mountain Monarchs – wild sheep and goats of the Himalaya. The University of Chicago Press
[11] Schäfer, Ernst, 1937: Unbekanntes Tibet – Durch die Wildnisse Osttibets. Paul Parey, Berlin.
[12] Shackleton, D. M (ed.) and the IUCN/SSC Caprinae Specialist Group, 1997: Wild Sheep and Goats and their Relatives. Status Survey and Conservation Action Plan for Caprinae. IUCN, Gland, Switzerland and Cambridge, UK. 390 + vii pp
[13] Smith, Andrew T. and Xie, Yan, 2008: A guide to the mammals of China. Princeton University Press
[14] Valdez, Raul, 1982: The wild sheep of the world. Wild Sheep and Goat International, Mesila, New Mexico 88046
[15] Tan, S.; Zou, D.; Tang, L.; Wang, G; Peng, Q.; Zeng, B; Zhang, C.; Zou, F., 2012: Molecular evidence for the subspecific differentiation of blue sheep (Pseudois nayaur) and polyphyletic origin of dwarf blue sheep (Pseudois schaeferi). Genetica. 2012 Jun; 140 (4-6):159-67. Epub 2012 Jul 21.
[16] Wilson, D. E. and Mittermeier, R. A. [eds], 2011: Handbook of the mammals of the world. Vol. 2. Hoofed mammals. Lynx Edicions, Barcelona.








